Background
When an exposure appears to be associated with an outcome, the outcome may, in fact, be caused by the indication for which the exposure was used, or some factor associated with the indication. The apparent association between the exposure and the outcome is then said to be confounded by the indication, which is the true cause of the outcome. In some cases, the indication may mask the outcome.
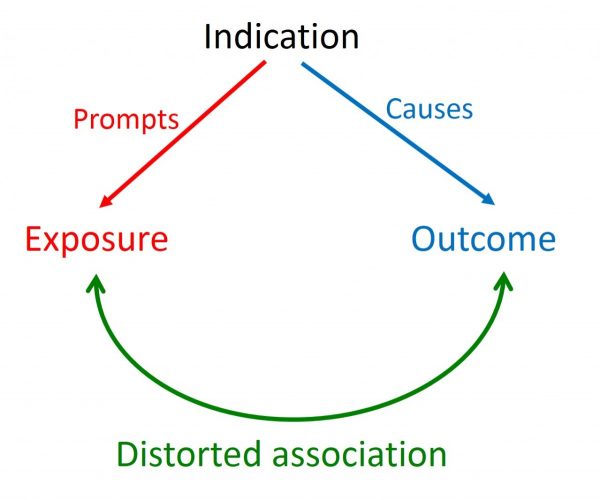
Figure 1. The general principle of confounding by indication
Confounding by indication is very common in observational studies (e.g. case-control and cohort studies). It can occur in relation to either beneficial outcomes or harmful outcomes and can result in either an increase or a reduction in the apparent risk of the outcome.
Example
An observational study suggested that children who had been given paracetamol were more likely to develop asthma, rhinoconjunctivitis, and eczema in later life; however, this result may have been confounded by an association between fever or infection and a later risk of asthma etc (Figure 2). Since this was a questionnaire study, recall bias could also have affected the result.
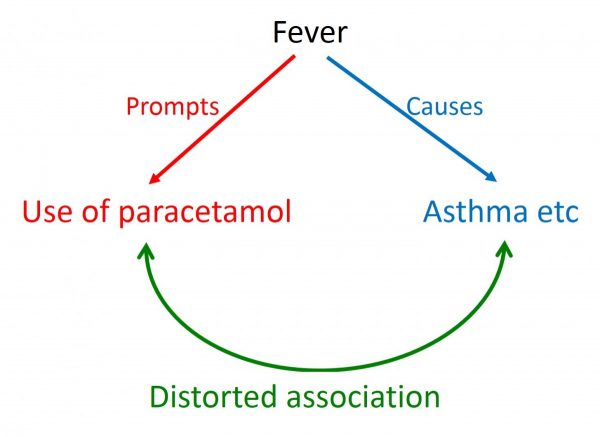
Figure 2. An example of potential confounding by indication of the observed increased risk of asthma in later life in children who were given paracetamol
A retrospective observational study concluded that angiotensin receptor blockers (ARBs) were more effective than angiotensin-converting-enzyme (ACE) inhibitors across a wide spectrum of cardiovascular diseases and on a number of different clinical outcomes, including cardiovascular mortality, non-fatal myocardial infarction, non-fatal stroke, or hospitalisation for cardiovascular disease. Despite using propensity scoring to match groups, it is plausible that a large number of patients who took an ACE inhibitor soon after a myocardial infarction (at high risk of adverse outcomes) were matched with patients who took an ARB for hypertension (at relatively lower risk) with similar propensity, presenting a high risk of confounding by indication.
Impact
In a systematic review of observational studies of the effectiveness of influenza vaccine, Remschmidt and colleagues investigated the impact of confounding by indication. Some studies showed that vaccine uptake was less likely in patients with chronic diseases, who were also less well, and therefore had a higher baseline risk of adverse health outcomes than healthy subjects. Unless adequate adjustment was made for the people who were less well when they received the vaccine, vaccine effectiveness may have been underestimated. Of the included 23 studies, the authors identified 17 (74%) that contained some form of confounding by indication. Twelve studies that reported all-cause mortality included adjustments for potential confounders (e.g. age, race, sex, comorbidities). Adjusting for confounders increased the beneficial effect of vaccination on all-cause mortality by 12% (95% CI: 7–17%).
The opposite effect (i.e. an overestimate of the size of the beneficial effect) can occur when participants who are vaccinated in a vaccine study have a lower prevalence of comorbidities, as indicated by baseline characteristics, than participants who are not vaccinated. Remschmidt and colleagues termed this “healthy vaccinee bias”, which is an example of volunteer bias.
Preventive steps
Careful study design can reduce the risk of confounding by indication.
For example, including a range of different indications for the same exposures enables the relationship between the exposure and the outcome in relation to each of the individual indications to be analysed separately. A consistent outcome across all indications will suggest that the outcome is indeed due to the exposure, since it is unlikely that each different indication would cause the same outcome.
This method was used in an observational study in which there was an increased incidence of oesophageal cancer in people taking proton pump inhibitors (PPIs); in this case the potential confounder was the indication for treatment (the reason for using the PPIs). The observed outcome, an increased risk of oesophageal cancer, may have resulted from either the treatment or the indication for the treatment. To investigate this, Brusselaers and colleagues analysed their data by separately considering (a) indications with an increased risk of cancer, (b) indications with no known association, and (c) indications with a reduced risk. The analyses showed a persistent relationship across all three groups, suggesting a true association between PPIs and oesophageal cancer.
Designing the study to include different indications allowed the authors to stratify by different indications and to conclude from the interpretation of the results that the exposure rather than the indication was more likely to have caused the outcome.
In interpreting the results of observational studies one should always be aware of the possibility of confounding by indication and take care to consider it in one’s interpretation. Results can be analysed keeping in mind known confounders related to the indication, controlling statistically for their effects. However, self-evidently, one cannot control for unknown confounders.

