Background
Insensitive-measure bias occurs when the method or tool or test used to detect the outcome of interest lacks accuracy. In compiling his 1979 list of biases in research, David Sackett defined insensitive-measure bias as: “When outcome measures are incapable of detecting clinically significant changes or differences, Type II errors occur.”
A Type II error is one in which a true association is not detected and genuine differences between groups are missed. Porta’s definition of Type II error: “The error of failing to reject a false test hypothesis; e.g., in null testing, declaring that a difference does not exist when in fact it does.”
Insensitive-measure bias can be considered a subtype of information bias.
Example
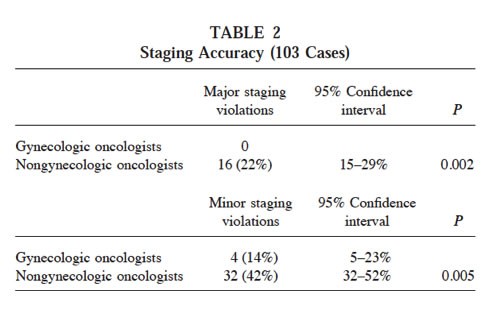
A US examined the hypothesis that tumour staging in the National Cancer Data Base (NCDB)/Surveillance Epidemiology and End Results (SEER) was not accurate enough to detect important clinical differences between patients.
Focusing on two common gynaecological cancers (ovarian and womb cancers), the authors state: “Insensitive-measure bias would exist if non-gynaecologic oncologists inaccurately staged gynecologic cancers compared with gynecologic oncologists.”
To determine if insensitive-measure bias was affecting results obtained by using SEER data, they examined the rate of “staging violations” (errors in the assessment of the staging of tumours) in their data records, as recorded by oncologists specialising in gynaecological cancers and by oncologists not specialising in gynaecological cancers:
Fanning J, Gangestad A, Andrews SJ. National Cancer Data Base/Surveillance Epidemiology and End Results: potential insensitive-measure bias. Gynecol Oncol. 2000 Jun;77(3):450-3
The authors concluded that the level of misclassification of tumour stage was too high for the data to be used reliably to examine hypotheses about causes of cancer or prognosis, due to insensitive-measure bias.
Impact
We have not found any studies assessing the impact of insensitive measure bias. The impact of information bias is considered in our catalogue entry for information bias.
Preventive steps
To obtain reliable results from medical studies it is important to use outcome assessment tools that are sufficiently sensitive to detect important clinical differences in the factors being investigated. It is also essential to use validated scale measures that have been shown to be sensitive to change and provide clinically important differences.

